Our laboratory research is focused on tumor therapy and can be divided into several interconnected areas:
-
Experimental Therapy – an interdisciplinary approach to studying the antitumor effects of newly synthesized compounds, as well as other natural and commercial potential anticancer agents.
-
Molecular Oncology: a) investigation of the role of ion transport, particularly potassium, in tumor treatment, tumor cell sensitivity to therapy, and invasivness; b) investigation of the role of adhesion and extracellular matrix modulation in tumor invasiveness and treatment.
Ad 1. Experimental Therapy.
We investigate the effects and/or mechanisms of action of newly synthesized compounds and other natural or commercial agents with potential anticancer activity. In collaboration with numerous research groups in organic chemistry from Croatia and abroad, we conduct screening studies of novel compounds, as well as detailed investigations of the mechanisms of the most active ones. Recently, our research has focused on the modulation of ABC transporters as a strategy to increase the sensitivity of highly resistant tumors to therapy.
This strongly interdisciplinary approach has resulted in unique expertise in medicinal/pharmaceutical chemistry, publications in respected journals in the field of medicinal chemistry, patents, and the discovery of several lead molecules.
Selected Publications and Patents:
a) Hranjec, M; Kralj, M*; Piantanida, I; Sedić, M; Šuman, L; Pavelić, K; Karminski-Zamola, G*. Novel cyano- and amidino-substituted derivatives of styryl-2-benzimidazoles and benzimidazo[1, 2-a]quinolines. Synthesis, photochemical synthesis, DNA binding, and antitumor evaluation, part 3. J Med Chem. 50 (2007) , 23; 5696,
b) M. Marjanović, M. Kralj*, F. Supek, L. Frkanec, I. Piantanida, T. Šmuc, Lj. Tušek-Božić. Antitumor potential of crown ethers: structure activity relationships, cell cycle disturbances and cell death studies of a series of ionophores. J Med Chem (2007) 50:1007-1018
c) Supek, F; Šumanovac Ramljak, T; Marjanović, M; Buljubašić, M; Kragol, G; Ilić, N; Šmuc, T; Zahradka, D; Mlinarić-Majerski, K*; Kralj, M*. Could LogP be a principal determinant of biological activity in 18-crown-6 ethers? Synthesis of biologically active adamantane-substituted diaza-crowns. Eur J Med Chem. 46 (2011) 3444-3454
d) Kralj, M*; Tušek-Božić, Lj; Frkanec, L. Biomedical Potentials of Crown Ethers: Prospective Antitumor Agents. ChemMedChem. (2008) 3:1478-1492
e) Ester, K; Hranjec, M; Piantanida, I; Ćaleta, I; Ivana, J; Pavelić, K; Kralj, M*; Karminski-Zamola, G*. Novel Derivatives of Pyridyl-Benzo[b]thiophene-2-carboxamides and Benzo[b]thieno[2, 3- c]naphthyridin-2-ones: Minor Structural Variations Provoke Major Differences of Antitumor Action Mechanisms. J Med Chem. 52:2482-2492 (2009).
f) Supek, F#; Kralj, M#; Marjanović, M; Šuman, L; Šmuc, T; Krizmanić, I; Žinić, B. Atypical cytostatic mechanism of N -1-sulfonylcytosine derivatives determined by in vitro screening and computational analysis. Invest New drugs. (2008) 26:97-110., #First autor – equal conribution
g) Perin, N; Martin Kleiner, I; Nhili, R; Laine, W; David-Cordonnier, M-H; Vugrek, O; Karminski-Zamola, G; Kralj, M*; Hranjec, M*. Biological activity and DNA binding studies of 2-substituted benzimidazo[1, 2-a]quinolines bearing different amino side chains. MedChemComm. 4 (2013) 1537-1550.
h) Basarić, N; Mlinarić-Majerski, K; Kralj, M. Quinone methides: photochemical generation and its application in biomedicine. Current Organic Chemistry. (2014) 18; 3-18.
i) Kralj, M*; Uzelac, L; Wang, Yu-H; Wan, P; Tireli, M; Mlinarić-Majerski, K; Piantanida, I; Basarić, N*. Enhancement of antiproliferative activity by phototautomerization of anthrylphenols. Photochem. Photobiol. Sci. (2015) 14:1082-1092.
j) Levatić, J; Ćurak, J; Kralj, M; Šmuc, T; Osmak, M; Supek, F. Accurate Models for P-gp Drug Recognition Induced from a Cancer Cell Line Cytotoxicity Screen. J Med Chem. 56 (2013) 5691-5708.
k) Patent No. US 8,389,505 B2 (2013): Marijeta Kralj, Kata Majerski, Marko Marjanović i Tatjana Šumanovac Ramljak „Adamantane derivatives of aza-crown ethers and their use in treatment of tumor“
l) Guberović I#, Marjanović M#, Mioč M, Ester K, Martin-Kleiner I, Šumanovac Ramljak T, Mlinarić-Majerski K, Kralj M. Crown ethers reverse P-glycoprotein-mediated multidrug resistance in cancer cells. Sci Rep, 14467 (2018). #First autor – equal conribution
m) Patent No. US11103519B2 / EP3612538B1 (2017): Basarić, Nikola; Kralj, Marijeta; Mikecin, Ana-Matea; Cindrić, Matej: Quinone-methide precursors with bodipy chromophore, method of preparation, biological activity and application in fluorescent labeling.
n) Zlatić K, Antol I*, Uzelac L, Mikecin Dražić A-M, Kralj M*, Bohne C, Basarić N*. Labeling of Proteins by BODIPY-Quinone Methides Utilizing Anti-Kasha Photochemistry. ACS Appl. Mater. Interfaces (2020), 12, 347−351
o) Mioč M, Telbisz Á, Radman K, Bertoša B, Šumanovac T, Sarkadi B, Kralj M*. Interaction of crown ethers with the ABCG2 transporter and their implication for multidrug resistance reversal. Histochem Cell Biol. (2022) 158 (2022) str. 261-277.
p) Zlatić, K; Popović, M; Uzelac, L ; Kralj, M*; Basarić, N*. Antiproliferative activity of meso-substituted BODIPY photocages: Effect of electrophiles vs singlet oxygen. Eur J Med Chem, (2023) 259 ; 1-15
q) Mioč M, Beus M, Carević K, Rajić Z, Sarkadi B, Telbisz Á*, Kralj M*. Selective Inhibition of the ABCG2 Transporter by Primaquine Derivatives Reverses the Multidrug Resistance of Tumor Cells. Int J Mol Sci. (2025) 26:5367.
Ad 2. Molecular Oncology
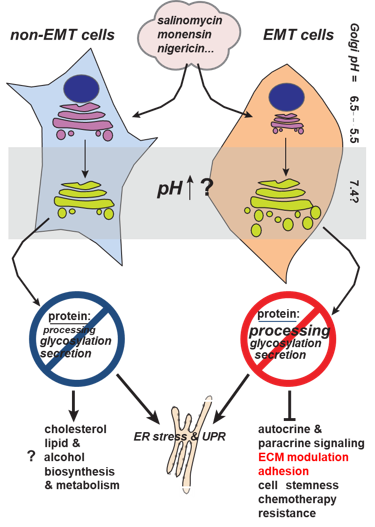
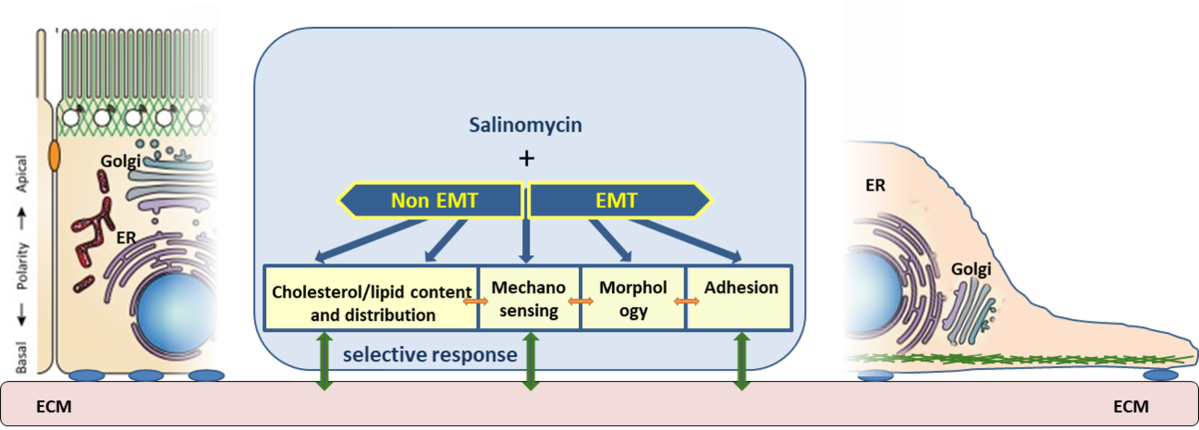
Our research has been focused on the potential antitumor activity of potassium ionophores. In particular, we have concentrated on the biological activity of crown ethers, which are also capable of transporting potassium across membranes, as well as on natural ionophores such as salinomycin, monensin, and nigericin. For example, the potassium ionophore salinomycin has been recognized as a potentially selective agent for the elimination of cancer stem cells (CSCs). Among their other characteristics, CSCs exhibit pronounced resistance to all forms of therapy. Consequently, our studies have aimed at discovering new compounds with potential selectivity for CSCs, as well as at investigating CSC properties.
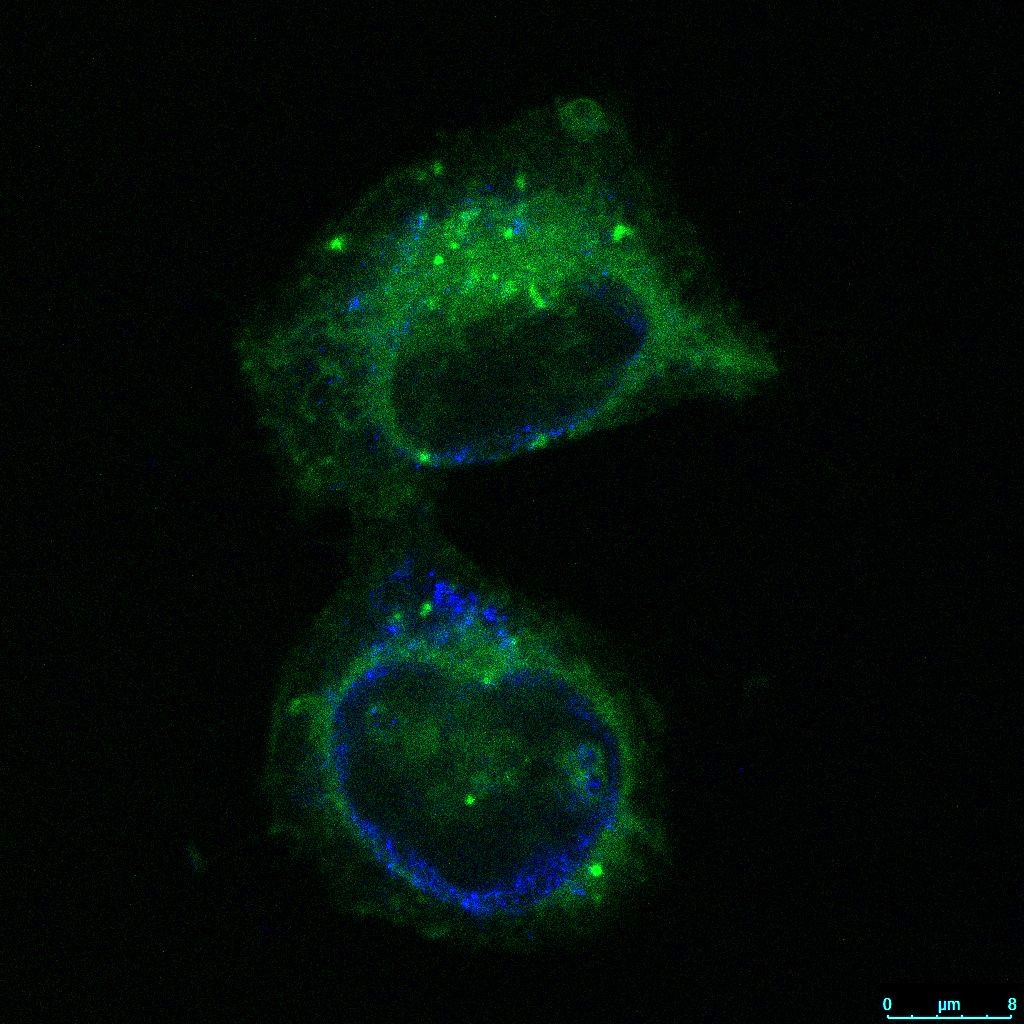
In our work, we use cell models that have undergone epithelial-to-mesenchymal transition (EMT) and thus display many CSC-like features. Within this framework, we investigate differences between EMT cells and their epithelial (non-EMT) counterparts. We have placed particular emphasis on elucidating the differential effects of salinomycin on various organelles in this and other cell models (Scientific Research Project “A multidisciplinary approach to discover selective drugs targeting cancer stem cells: The role of potassium transport – MultiCaST”, Croatian Science Foundation).
In our studies, we were the first to demonstrate that salinomycin affects the function and morphology of the Golgi apparatus, with stronger effects observed in EMT cells.
Further research based on global gene expression analysis revealed that adhesion is the most affected biological process in EMT cells after salinomycin treatment, a phenomenon not observed in non-EMT cells. These differential mechanisms are currently under investigation.
Selected Publications:
- Marjanović M#, Mikecin Dražić A-M#, Mioč M, Paradžik M, Kliček F, Novokmet M, Lauc G, Kralj M*. Salinomycin disturbs Golgi apparatus function and specifically affects cells in epithelial-to-mesenchymal transition. Journal of Cell Science. (2023)136(17):jcs260934.
- Ester, K. et al. Targeting plasma membrane and mitochondrial instability in breast cancer cells and breast epithelial to mesenchymal transition-model cells by adamantyl diaza-crown ether ZG613. bioRxiv 2022.09.30.510273 (2022) doi:10.1101/2022.09.30.510273.
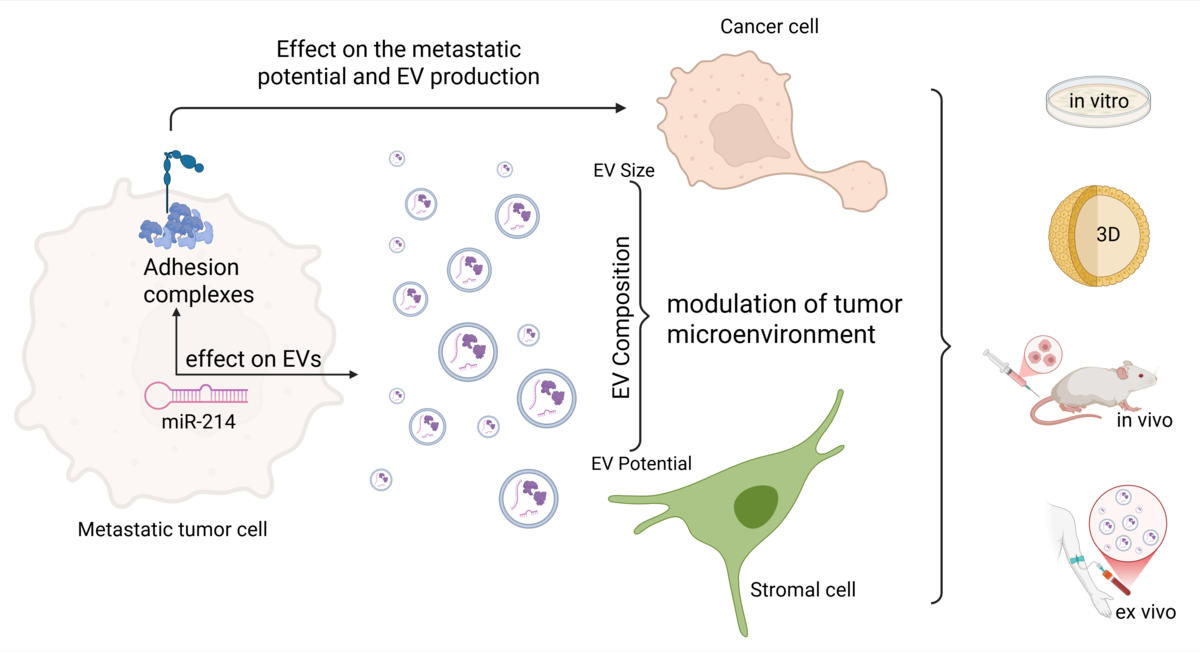
Research into adhesion and adhesion complexes is being continued using a model for triple-negative breast cancer. Particular attention will be paid to the role of small non-coding RNA molecules (miR) in the regulation of adhesion complexes, their contribution to increasing metastatic potential, their influence on the formation of extracellular vesicles as a means of intercellular communication, and, more generally, their effects on communication between tumour cells and cells of the microenvironment.
Selected Publications:
- Orso F, Virga F, Dettori D, Dalmasso A, Paradzik M, Savino A, Pomatto M.A.C., Quirico L, Cucinelli S, Coco M, Mareschi K, Fagioli F, Salmena L, Camussi G, Provero P, Poli V, Mazzone M, Pandolfi P P. & Taverna D. Stroma-derived miR-214 coordinates tumor dissemination J Exp Clin Cancer Res 2023 Jan 13;42(1):20. doi: 10.1186/s13046-022-02553-5
- Quirico L, Orso F, Cucinelli S, Paradžik M, Natalini D, Centonze G, Dalmasso A, La Vecchia S, Coco M, Audrito V, Riganti C, Defillipi P, Taverna D miRNA-guided reprogramming of glucose and glutamine metabolism and its impact on cell adhesion/migration during solid tumor progression; Cell Mol Life Sci. 2022 Mar 29;79(4):216. doi: 10.1007/s00018-022-04228-y.
The study of adhesion complexes has also led to interdisciplinary research (evolution, cell biology, bioinformatics) focused on the origin of the interactions between the components of the cytoskeleton (actin filaments, microtubules, intermediate filaments). In this work, we have analysed the evolutionary conservation of these interactions, and identified key proteins that enable these interactions in silico.
Selected Publications:
- Paradžik T, Podgorski I I, Vojvoda Zeljko T, Paradžik M*. Ancient Origins of Cytoskeletal Crosstalk: Spectraplakin-like Proteins Precede the Emergence of Cortical Microtubule Stabilization Complexes as Crosslinker; Int J Mol Sci. 2022 May 17;23(10):5594. doi: 10.3390/ijms23105594.
Previous research:
Our previous research has focused on the complex regulatory networks controlling cell cycle progression and cell death in tumor cells, with the aim of understanding how anticancer agents induce cell death and how defects in these pathways affect therapy outcomes. Particular attention was given to the p53 gene, the key regulator of most treatment-related cellular responses and one of the most frequently mutated genes in human cancers, as well as the p21WAF1/CIP1 gene, which plays a central role in senescence, but has a contradictory function in apoptosis and an underexplored role in autophagy.
We investigated the correlation between specific DNA lesions induced by antitumor compounds and the resulting cellular responses, emphasizing the importance of non-apoptotic mechanisms such as senescence, mitotic catastrophe, and autophagy, and their interplay with apoptosis. Additionally, we studied how the p21 gene regulates different cellular responses and whether and how p21 status influences tumor cell chemosensitivity.
Taken together, these studies have provided important insights into the molecular determinants of therapy response, contributing to the identification of novel targets and strategies for more effective cancer treatment. (MSES project “The role of different cell death responses to DNA-damage treatment” (2007-2013))
- Kraljević Pavelić, S; Čačev, T; Kralj, M. A dual role of p21waf1/cip1 gene in apoptosis of HEp-2 treated with cisplatin or methotrexate. Cancer Gene Ther. (2008) 15:576-590.
- Kraljević Pavelić, S#; Marjanović, M#; Poznić, M; Kralj, M. (2009) Adenovirally-mediated p53 overexpression diversely influence the cell cycle of HEp-2 and CAL 27 cell lines upon cisplatin and methotrexate treatment. J Cancer Res Clin Oncol. 135:1747-1761. #First autor – equal conribution
- Mikecin A-M, Walker L, Kuna M, Raucher D. Thermally targeted p21 peptide enhances bortezomib cytotoxicity in androgen-independent prostate cancer cell lines. Anticancer Drugs. (2014) 2: 189–199.