Asymmetric desymmetrization
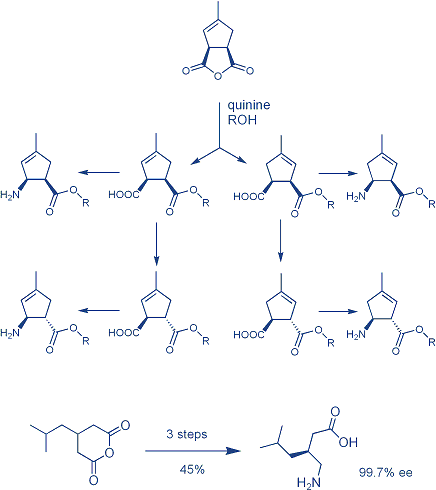
Asymmetric desymmetrization (ADS) of relatively simple meso compounds can provide highly practical access to a variety of usefully functionalized chiral building blocks for the asymmetric synthesis of numerous targets. One of the best examples is the ADS of cyclic meso-anhydrides catalyzed by natural alkaloids or modified alkaloids, since the resulting functional groups can selectively be manipulated in further reaction steps. Thus, starting from easily available succinic and glutaric anhydrides a variety of enantiomerically pure β- and γ-aminoacids can be prepared; some of them already being biologically active (Icofungipen, Pregabalin).
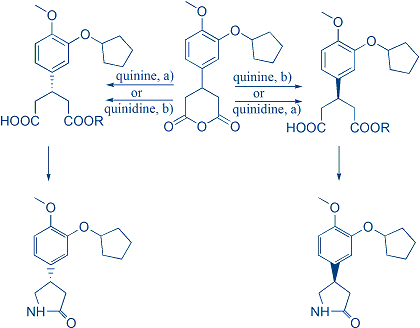
Continuing the research, an unexpected inversion of enantioslectivity depending on reaction conditions was observed, allowing for the utilization of one catalyst for both product enantiomers. After thorough research the mechanism of reaction was proposed, and application demonstrated in the synthesis of Rolipram.
Synthesis and transformations of chiral aziridines
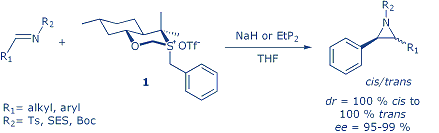
Chiral aziridines are highly valuable synthetic intermediates because of their ability to undergo different transformations, especially ring opening, to afford versatile optically active nitrogen-containing compounds. Amongst different methods for asymmetric aziridinations the large area belongs to imine aziridinations and includes: aza-Darzens reactions, and reactions of imines with carbenes or sulfur ylides. Our work here is focused on the chiral sulfur ylide mediated asymmetric imine aziridinations.
A series of 2,3-disubstituted N-Ts, N-Boc, and N-SES aziridines were synthesized using sulfonium salt 1derived from Eliel’s oxathiane.Both phosphazene base EtP2, and sodium hydride were used as bases to generate the ylide. The two-step synthesis provided in most casescis/trans mixtures of aziridines with both isomers having high enantiomeric purity (>95 %). Diastereoselectivity of this reaction depends on the nitrogen protecting group, and on the imine substituent. Further investigations of the influence of ring-substituent on diastereoselectivity are ongoing.
Selected publications:
T. Ivšić, I. Dokli, A. Rimac, Z. Hameršak, “Synthesis of GABOB and GABOB-Based Chiral Units Possessing Distinct Protecting Groups”, Eur. J. Org. Chem. 2014 (2014) 631–638.
T. Ivšić, and Z. Hameršak, "A simple enantioselective route toward (R)- and (S)-Rolipram via anhydride desymmetrization", Tetrahedron: Asymmetry 24 (2013) 217–222.
- T. Ivšić, J. Novak, N. Došlić, and Z. Hameršak, "One catalyst for both enantiomers: uncovering the inversion of enantioselectivity in cinchona-mediated desymmetrization of glutaric meso-anhydrides", Tetrahedron 68 (2012) 8311–8317.
- I. Dokli, I. Matanović, and Z. Hameršak, ″Sulfur Ylide Promoted Synthesis of N-Protected Aziridines: A Combined Experimental and Computational Approach″, Chem. Eur. J. 16 (2010) 11744-11752.
- T. Ivšić, and Z. Hameršak, "Inversion of enantioselectivity in quinine-mediated desymmetrization of glutaric meso-anhydrides", Tetrahedron: Asymmetry 20 (2009) 1095-1098.
- I. Stipetić, M. Roje, and Z. Hameršak: “Highly Enantioselective Aziridination of N-Protected Imines: Comparison of the Phosphazene EtP2 and Sodium Hydride as Bases”, Synlett (2008) 3149-3152.
- Z. Hameršak, I. Stipetić, and A. Avdagić, “An Efficient Synthesis of (S)-3-Aminomethyl-5-methylhexanoic Acid (Pregabalin) via Quinine-Mediated Desymmetrization of Cyclic Anhydride”, Tetrahedron: Asymmetry 18 (2007) 1481-1485.
- Z. Hameršak, M. Roje, A. Avdagić, and V. Šunjić, “Quinine-Mediated Parallel Kinetic Resolution of Racemic Cyclic Anhydride: Stereoselective Synthesis, Relative and Absolute Configuration of Novel Alicyclic γ-Amino Acids”, Tetrahedron: Asymmetry 18 (2007) 635–644.