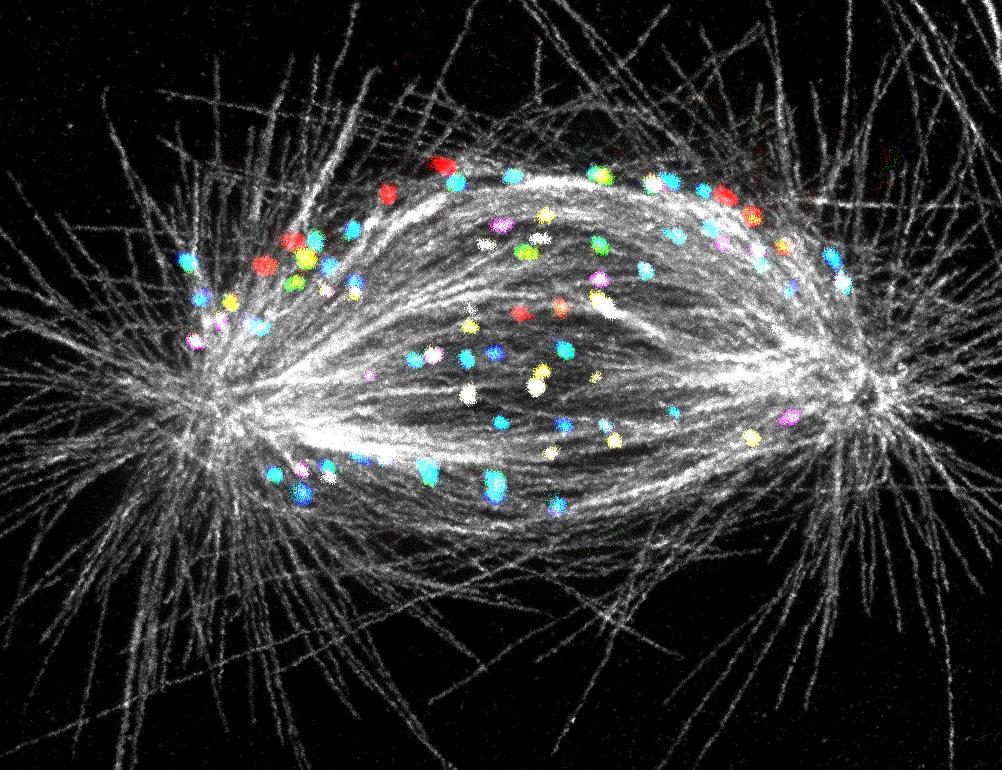
In their latest study, published today in the prestigious scientific journal Nature Communications, a team of molecular biologists from the RBI shows how the cell corrects one of the riskiest situations during division, when a chromosome at the beginning of division ends up behind the pole of the mitotic apparatus, in a location from which it is more difficult to guide it correctly.
When a Chromosome Gets Stuck in an Unfavorable Position
When a single cell divides into two new ones, it must distribute its chromosomes, the carriers of genetic instructions, with great precision. In most cases, this process proceeds smoothly, but sometimes a chromosome ends up in a particularly unfavorable position, behind the pole of the mitotic apparatus, in a kind of “blind spot”.
From this position, the chromosome is less accessible to the mechanisms that normally ensure proper chromosome distribution between the two new cells. Such chromosomes therefore represent a higher risk for division errors. If they fail to escape this unfavorable position in time, they may end up in the wrong cell or lag behind during division, as this research team demonstrated in a study published in Nature four years ago. The new research brings an encouraging answer, showing that the cell has a rescue mechanism for such situations.
“In this work, we showed that there is a particularly sensitive moment at the beginning of division, when a chromosome can end up in a position from which it is difficult to distribute it correctly. At the same time, we demonstrated that the cell has a way to resolve this problem. For the chromosome to be pulled out of this ‘blind spot’, the mitotic apparatus must elongate sufficiently, thereby opening a path toward proper division,” explains Prof. Iva Tolić.
In a Rescue Mission, the Cell Reorganizes the Entire System
The key finding of the study is that rescuing such chromosomes does not depend only on whether the cell can “capture” them, but on whether it can reorganize the entire mitotic apparatus in time.
“As the poles of the mitotic spindle move apart, the fibers connected to the problematic chromosome change their position and pull it out of the zone of highest risk. Only once the chromosome has moved into a more favorable region of the mitotic apparatus can the cell guide it more reliably toward proper distribution,” explains Dr. Isabella Koprivec, the first author of the study.
This shows that the precision of cell division does not depend solely on the initial arrangement, but also on the cell’s ability to respond to a problem and reorganize its internal structure during division.
“This insight is important because it shows that the error is not necessarily in the chromosome itself, but also in how effectively the cell can reorganize at the right moment. In some tumor cells, this process is less efficient, so chromosomes remain longer in positions where they are more prone to errors,” says Dr. Valentina Štimac, co-first author of the study.
The forming mitotic spindle captured by Isabella Koprivec and Valentina Štimac
Cell Division Observed Live
The scientists reached these conclusions through an innovative combination of several advanced methods for observing cells. They monitored cell division live, in real time, which allowed them to see where chromosomes are located, how they move, and what happens when some of them end up in risky positions. They also used very high-resolution microscopy to study the finest structural details that govern chromosome movement. In this way, they connected the sequence of events over time with precise insights into the fine structures that enable this process.
The mechanism was first analyzed in detail in model human cells, and then examined in several tumor cell lines. The same issue was observed there as well, but its resolution was slower when the mitotic apparatus failed to elongate efficiently.
The scientists therefore selectively modified the activity of proteins that influence how much the mitotic apparatus can elongate. When elongation was weaker, chromosomes were extracted more slowly from the risky position. When elongation was stronger, the process was faster.
An Important Step in Understanding Errors in Tumor Cells
The discovery is important because it offers a new and concrete explanation of how the cell attempts to prevent errors in chromosome distribution. Such errors are a major source of genetic instability, which is frequently present in tumor cells. The research helps to better understand why some cells are more prone to improper chromosome distribution and what happens when the mechanisms that should correct these errors are not sufficiently effective.
“Cell division must be extremely precise, and this work shows how dynamic that precision actually is. The cell constantly monitors the situation and adapts, even when things do not proceed ideally at the start. It is precisely within these adaptive mechanisms that part of the answer lies to how the stability of genetic material is maintained, and what happens when this system fails,” concludes Prof. Iva Tolić.
The research was conducted entirely at the Ruđer Bošković Institute. Alongside Prof. Iva Tolić, the main authors are former PhD students Isabella Koprivec and Valentina Štimac, who have since continued successful careers, Isabella in medical and scientific communication, and Valentina as a postdoctoral researcher at a renowned institute in Bonn. Other contributors to the project include postdoctoral researcher Dr. Kruno Vukušić, PhD student Mario Đura, and former graduate student Petra Mikec, now enrolled in a prestigious doctoral program in Marseille.
The research was funded by the Croatian Science Foundation through the program “Scientific Cooperation” for collaboration with Croatian scientists in the diaspora, by a European Research Council Synergy project, and by the IPSted and QuantiXLie Centre of Excellence projects, co-financed by the Government of the Republic of Croatia through the European Regional Development Fund. An important role was also played by the infrastructure of the University Computing Centre, SRCE, whose technological support enabled the execution of this complex research.